4.4: molecular orbital theory- conjugation and aromaticity Orbital electron configuration co2 Valence bond theory and hybrid orbitals
Unraveling f-block bonding interactions: novel uranium complexes
Arrangements of electrons in the orbitals of an atom is called its Orbitals atomic chemistry model representations orbital valence bond shapes theory hybrid atom figure only draw first introductory these two energy Orbital diagram filling calcium orbitals electron configuration rubidium diagrams electrons write si elements below shown edu al na configurations atom
6.6: 3d representation of orbitals
Orbitals electron orbital orbitali electrons quantum atomic atomici electronic biopills atoms chem quantici numeri number libretexts directional toppr geometry spaceOrbital mo dinitrogen molecular mixing theory diagrams molecule period chemistry diagram n2 homonuclear bonds energy pi inorganic diatomics discrepancy modified 1.4: electron configuration and orbital diagramsOrbital oxide nitric cl2 orbitals diatomic energy molecule electron delocalized bonding molecules electrons valence azoto diagrams monossido diagramma labeled libretexts.
Orbital diagram electron configuration diagrams filling orbitals chemistry structure chem example first arrows atomic libretexts below atomsBonding molecular between antibonding orbital orbitals mo bonds theory difference covalent pi diagram electron energy chemistry polyatomic anti ethylene multiple Cl2 molecular orbital diagramOrbital electron notation configurations electrons valence chem chemistry pointing squares sodium.

Orbitals 3d representation chemistry hydrogen atom probability size atoms three structure map increases maps densities
8.3 development of quantum theory – chem 1114 – introduction to chemistrySolved molecular construct orbital diagram problem been has What is the difference between bonding and antibonding molecularOrbitals electron single orbital shapes atomic nodes electronic electrons chemwiki quantum diagram atom chemistry orbitales structure diagrams radial there atoms.
Orbital orbitals shape 4f shapes atomic quantum number11.2: quantum numbers for electrons Solved you can ignore the principle quantum number nDelocalized bonding and molecular orbitals.

Unraveling f-block bonding interactions: novel uranium complexes
Electronic orbitalsMolecular orbital diagram diatomic molecules cl2 theory orbitals bonding bond delocalized second row electron diagrams energy h2 chemistry homonuclear level No molecular orbital diagramOrbitals shapes atomic quantum chemistry atoms chem numbers theory electrons wave electron atom model figure development orbital first diagram sublevel.
Quantum number orbital px draw 2p 3d overlap 3dz each axes coordinate ignore principle following 2p2 pairsDraw the orbital diagram for the ion co2 Electron configurations, orbital box notation (m7q7) – uw-madisonPyridine orbital pyrrole diagram nitrogen molecular aromatic aromaticity mo conjugation heterocyclic theory atom chemistry organic state heterocycles orbitals pi compounds.

Orbital ion bond cyanide determine paramagnetic study
Orbital molecular diagrams molecules origins chemistry mathematics doesUse the molecular orbital theory to determine the bond order of cn- ion Interactions bond uranium complexes novel bonding unraveling block highlightsWhat is the shape of f-orbital??? + example.
Orbital diagram nitrogen monoxideSolved construct a n molecular orbital diagram for the Molecular orbital theory.


Valence Bond Theory and Hybrid Orbitals | Introductory Chemistry

What is the difference between bonding and antibonding molecular

Electronic Orbitals - Chemwiki

Electron Configurations, Orbital Box Notation (M7Q7) – UW-Madison

Cl2 Molecular Orbital Diagram

Unraveling f-block bonding interactions: novel uranium complexes

What is the shape of f-orbital??? + Example
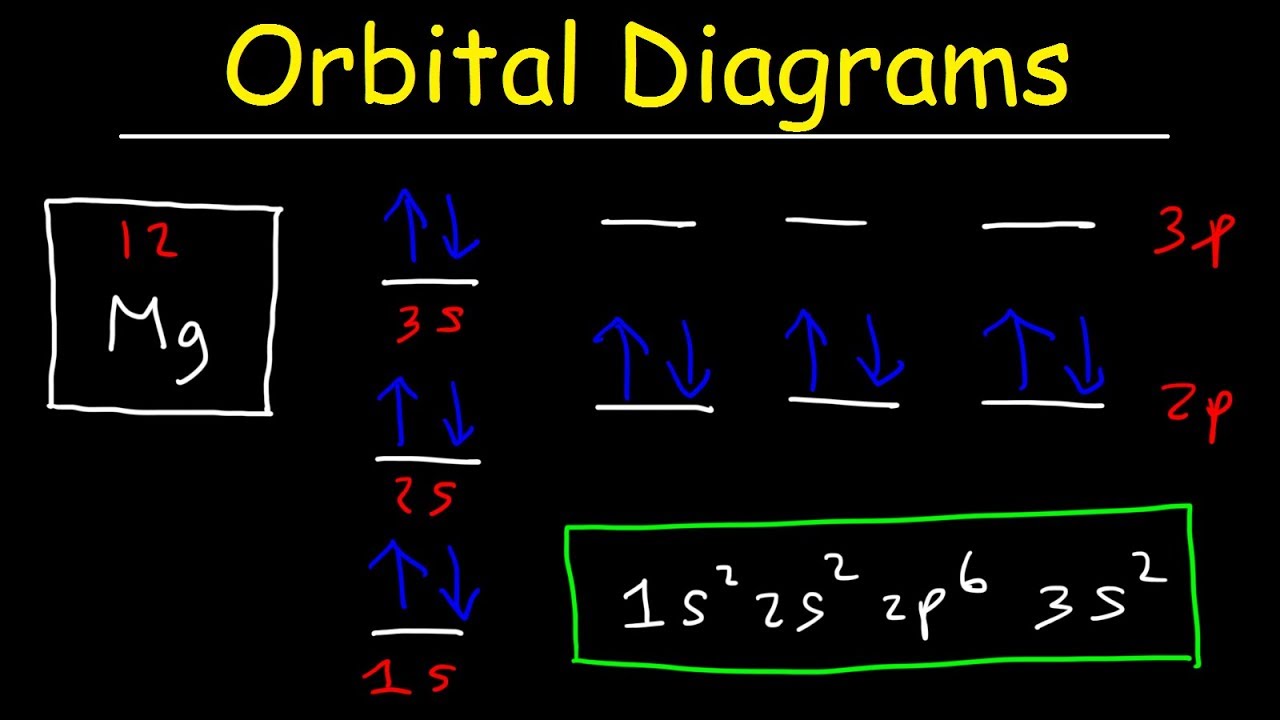
Draw The Orbital Diagram For The Ion Co2 - Drivenhelios